Most chemical reactions discussed in general chemistry occur in gaseous or aqueous phases. Here, an aqueous solution refers to a solution where water is the solvent. Although water molecules are overall neutral, they possess polarity, which results in strong intermolecular interactions known as hydrogen bonds. For a substance to dissolve in water and disrupt the hydrogen-bonded interactions between water molecules, it must also exhibit polarity. If a molecule forces itself into water, it must have properties compatible with those of water molecules to be accepted, meaning that polar solutes, which share similar characteristics with water, are more likely to dissolve in it. In contrast, low-polarity molecules, such as lipids, do not interact well with water molecules. As the saying goes, “oil and water don’t mix” — substances with different properties cannot easily associate. As a result, nonpolar substances in water are repelled by water molecules, causing lipid molecules to clump together rather than interact with water. This illustrates an important chemical principle: “Like dissolves like.” While this concept is so intuitive that it may seem redundant to explain, articulating it in detail can be quite challenging. Let’s dive into this complex topic.
In introductory chemistry courses, we learn that the dissolution of polar substances in water is driven by an increase in entropy. However, in graduate studies, as we delve into solvation shells, enthalpy becomes a more significant consideration. Similarly, while undergraduate courses teach that the hydrophobic effect of nonpolar molecules is primarily due to entropy increase, in advanced studies, enthalpy’s role is acknowledged as substantial in hydrophobic interactions. While this may seem perplexing, chemistry often involves seemingly contradictory concepts — one of my favorites is the term “unstable yet stable (relatively),” which we often use in chemistry.
When asked about enthalpy and entropy, most scientists know they are state functions that define Gibbs free energy, represented by the formula ΔG=ΔH−TΔS. However, summarizing each concept in a single sentence can be challenging. Among various explanations, the one I found most straightforward is this: Enthalpy is the “amount of heat and work (energy) absorbed or released when a substance forms (or changes) under constant pressure.” Since losing heat and work is easier than gaining them, reactions generally proceed in a direction where enthalpy decreases. Entropy, often referred to as “disorder,” is the “amount of heat that cannot be converted into work.” Work occurs when the movement of substances follows a specific direction. Therefore, as entropy increases, the movement of substances becomes more disordered. According to the second law of thermodynamics, the world tends toward increasing entropy. For a reaction to occur spontaneously, enthalpy must decrease (negative ΔH), and entropy must increase (positive ΔS), resulting in a negative change in Gibbs free energy (ΔG).
A complex question arises here: Strong intermolecular interactions restrict molecular movement, leading to decreased enthalpy. However, if these interactions are strong, entropy also decreases. This is like children trying to run freely in a playground but being restrained by holding hands with a buddy. Thus, scenarios where both enthalpy decreases and entropy increases are not easily imagined. In small-scale molecular reactions, stabilization by enthalpy generally outweighs the molecule’s degrees of freedom (entropy). However, in aqueous reactions, enthalpy and entropy often compete.
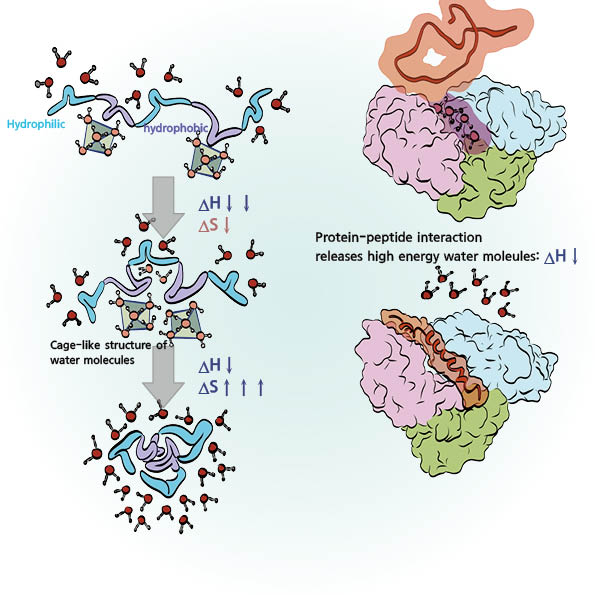
Hydrophobic interactions are crucial in protein folding and interactions. Proteins, as organic molecules based on carbon, consist of hydrocarbons (carbon bonded with hydrogen), which are representative nonpolar molecules. Additional elements like phosphorus (P), oxygen (O), nitrogen (N), and sulfur (S) attach to these hydrocarbons, creating polar functional groups. The combination of polar functional groups, which interact with water, and nonpolar functional groups, which repel water, results in complex interactions in aqueous environments. Within a protein, nonpolar functional groups avoid water by folding inward or clustering with other nonpolar groups, facilitating protein folding and interactions. Water molecules surrounding nonpolar groups in the protein form cage-like structures, reducing entropy. However, when nonpolar groups cluster or move inward, they release water, increasing degrees of freedom and entropy. While protein clustering reduces entropy within the protein, the larger increase in water molecules’ entropy results in an overall entropy increase. For this reason, protein folding and interactions are commonly explained as entropy-driven processes.
But what about enthalpy? Depending on the size and properties of the protein, enthalpy’s role can vary. The hydrophobic effect, where nonpolar interactions are essentially “pushed” by water, is a passive process, meaning the cause lies outside the protein. In contrast, van der Waals forces actively play a role. Van der Waals forces refer to intermolecular attractions or repulsions that are neither covalent bonds nor ionic interactions, existing between all materials. Their importance is heightened in nonpolar molecules without electrostatic interactions from polar groups. The effect of van der Waals forces increases with molecule size, which makes them especially significant in large protein complexes. Historically, the enthalpic aspect of hydrophobic interactions was limited to this.
The degree to which enthalpy and entropy influence protein folding varies with the stage of folding. Though not an actual structure, consider a fully extended protein as an example. As folding progresses, enthalpy initially decreases significantly, slowing as the structure becomes complex, even eventually increasing. Conversely, entropy decreases markedly in the early stages of folding but then increases as folding proceeds. Aside from initial entropic barriers, protein folding generally progresses due to entropy increase. Recent research indicates that enthalpy affects local interactions, known as docking, in proteins. Hydrophobic docking occurs in very confined spaces, where interestingly, a limited number of water molecules exist. These water molecules, restricted by the tight space, struggle to form proper hydrogen bonds. When a hydrophobic molecule or peptide binds in this region, water molecules, unable to form hydrogen bonds, are expelled and form stable hydrogen bonds in the bulk water, decreasing enthalpy. Furthermore, bonds forming between hydrophobic groups expose polar regions to water, enhancing interactions with water and further decreasing enthalpy. While such interactions are limited to local interactions, they are crucial for drug development targeting specific proteins.
Please visit the Hugh Kim Research Group homepage.
References
- Oroguchi, T., & Nakasako, M. Sci Rep 2016, 6 (1), 1-14
- Peter Atkins, P., & De Paula, J. Atkins’ physical chemistry. OUP Oxford, 2014
- Steinfeld, J. I., Francisco J. S., & Hase, W. L. Chemical kinetics and dynamics. Upper Saddle River, NJ: Prentice Hall, 1999
- Biedermann, F., Nau, W. M., & Schneider, H. J. Angew Chem Int 2014, 53 (42), 11158-11171
- Aksel, T., Majumdar, A., & Barrick, D. Structure 2011, 19 (3), 349-360

Leave a comment